
Mon - Sat 9:00 - 17:30

BROMINE SAFETY: HANDLING, STORAGE & TRASPORTATION
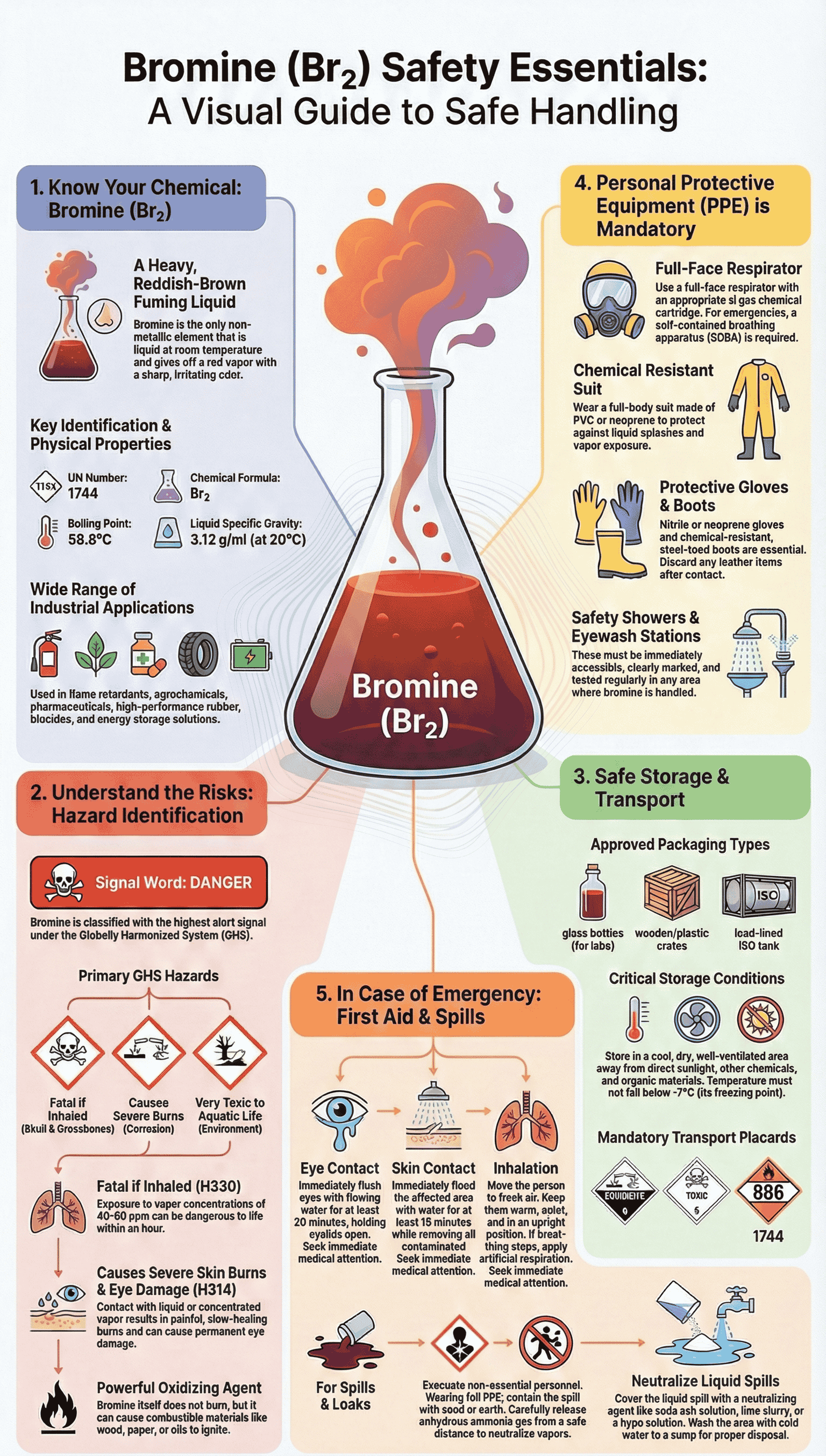
The story of Bromine is one of a "silent, heavy, and colorful" danger. Known for its distinct reddish-brown appearance, it is one of the few elements that is liquid at room temperature but readily evaporates into a suffocating gas.
In the industrial heartlands of India, particularly in the chemical belt of Gujarat, Bromine is a vital component for pharmaceuticals and flame retardants—but when safety protocols fail, it transforms into a localized disaster.
Bromine is a "heavyweight" among gases. Because its vapor is much denser than air, it does not rise and dissipate quickly; instead, it crawls along the ground, filling pits, trenches, and basements.
| Property | Danger Level | Effect |
| Physical State | Fuming Liquid | Readily turns into gas at room temperature ($20^circ ext{C}$). |
| Appearance | Red-Brown | Creates a visible "orange cloud" that signals danger. |
| Toxicity | High | Corrosive to the eyes, skin, and respiratory tract. Fatal if inhaled in high doses. |
| Reactivity | Oxidizer | Can cause organic materials (like wood or cloth) to catch fire spontaneously. |
One of the most significant recent bromine-related incidents in India occurred on August 23, 2023, at the PI Industries facility in Sarod village, Jambusar (Bharuch district, Gujarat).
At approximately 1:00 PM, a fire broke out in a storage tank containing bromine. The heat caused the liquid bromine to rapidly vaporize, and a massive plume of thick, orange-colored smoke began to hover over the factory and the surrounding village.
Mass Evacuation: Panic struck as the red fumes became visible for kilometers. Approximately 2,000 workers had to be evacuated immediately from the site.
Hospitalizations: At least 28 workers were rushed to the hospital after complaining of severe breathing difficulties, throat irritation, and nausea.
Environmental Panic: The "red sky" caused alarm among local residents, fearing long-term respiratory damage.
Preliminary investigations pointed toward a technical fault in the storage tank's cooling or pressure systems, which allowed the internal temperature to rise, leading to a fire and subsequent tank failure. The incident highlighted that even in modern facilities, the storage of "fuming" chemicals requires constant, fail-safe monitoring.
The Sarod incident and others like it serve as a grim reminder of the "3-S" rule for Bromine safety: Storage, Shielding, and Shift.
Cooling Systems: Bromine must be stored in cool, well-ventilated areas away from direct sunlight.
Secondary Containment: Always use glass-lined or lead-lined steel containers. Storage areas should have "dike" walls to catch leaks.
Scrubbers: Facilities must have alkaline scrubbers (using Caustic Soda) to neutralize any bromine vapors before they reach the atmosphere.
Standard cloth masks are useless against bromine.
Respiratory: Self-Contained Breathing Apparatus (SCBA) or full-face respirators with specific acid-gas cartridges.
Skin: Bromine is highly corrosive. Workers must wear Viton or Neoprene gloves and chemical-resistant suits.
Approach from Upwind: As seen in the Bharuch leak, the gas moves with the wind. Always move perpendicular to the wind or stay upwind (the wind should be blowing from your back toward the leak).
Water Spray: Use water curtains to "knock down" the vapor cloud, but avoid spraying water directly into a leaking liquid container, as this can cause a violent reaction.
Safety Note: If you see orange or reddish-brown fumes in an industrial setting, do not wait for an alarm. Evacuate immediately in the upwind direction.