
Mon - Sat 9:00 - 17:30

Alright, welcome to your first day on the floor. I'm your mentor for this training, and we're going to talk about chlorine. Before we even touch a cylinder, I want you to understand something. Chlorine is a paradoxical element. On one hand, it's a pillar of modern life—it purifies our drinking water, it's essential for manufacturing life-saving pharmaceuticals, and it's a key ingredient in everything from PVC pipes to computer chips.
On the other hand, it's so dangerous it was used as a chemical weapon. It demands absolute, unwavering respect.
Today isn't about memorizing a checklist of rules. It's about understanding the why behind every single step we take. The procedures we follow are written in the hard lessons of past incidents—lessons about what happens to human lung tissue when it's exposed to acid. Understanding the why is what keeps you safe. Let's start with the basics.
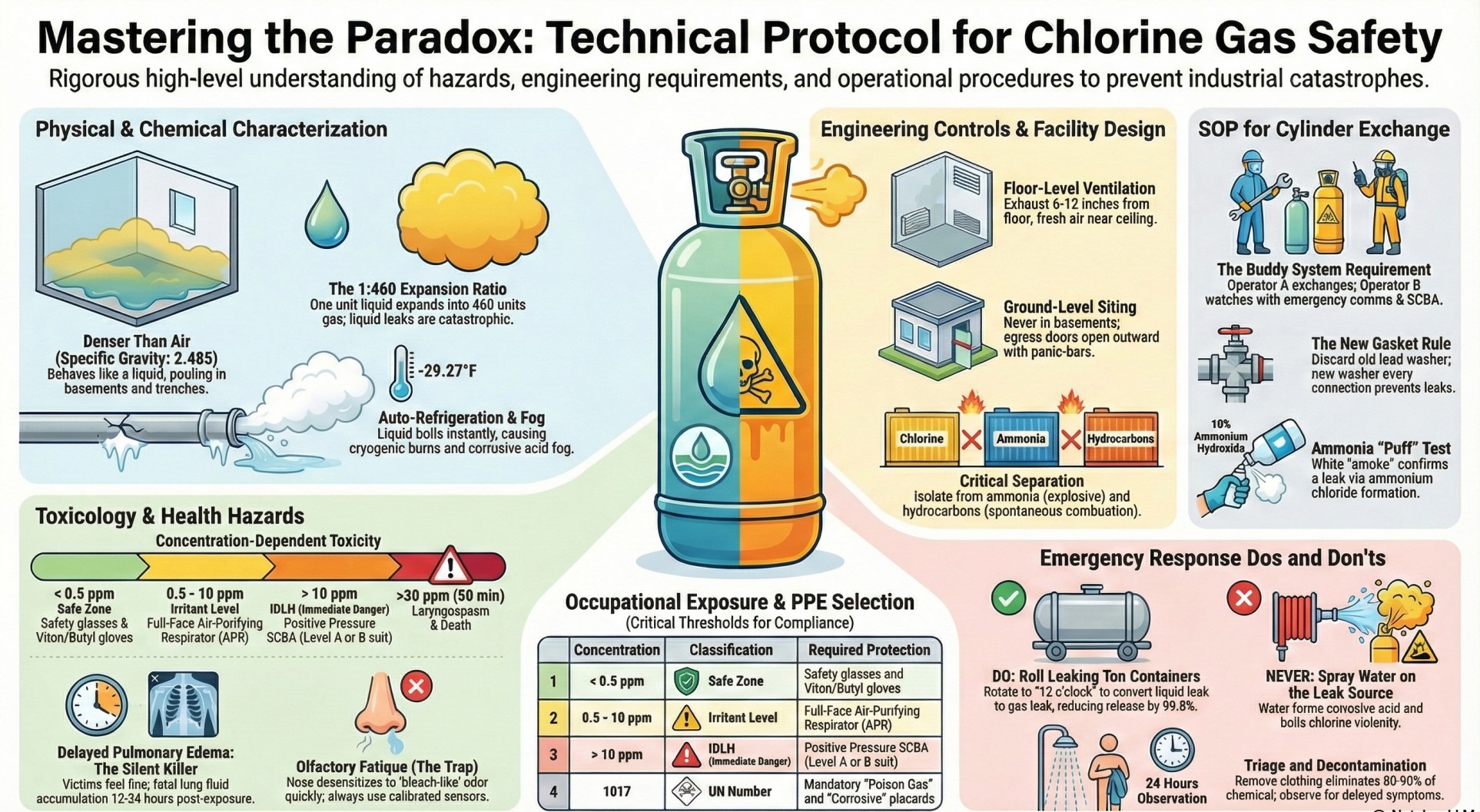
Before you handle chlorine, you need to internalize its fundamental nature. There are three truths you must never forget.
Now that you understand what we're dealing with, let's talk about the first and most important rule that has nothing to do with the chemical itself.
The Buddy System is non-negotiable. No one handles chlorine alone. Ever.
This rule exists because of chlorine's rapid, incapacitating effects. A high-level exposure doesn't just make you cough. It can cause an immediate laryngospasm—the violent constriction of your larynx that effectively strangles you. The intense irritation to your eyes can cause temporary blindness. In these conditions, self-rescue is nearly impossible.
Furthermore, chlorine can be a silent killer. A person can get a nasty exposure, feel better after a few minutes of fresh air, and then die 12 hours later as their lungs slowly fill with fluid. Your buddy is there to ensure you get immediate help and mandatory medical observation, no matter how you feel. They are your lifeline.
This is the most common task you'll perform. Let's walk through changing a 150-lb cylinder. Every step has a reason.
3.1. Gearing Up: The Right Armor
First, we put on our Personal Protective Equipment (PPE). We're not just grabbing any gloves and goggles.
3.2. Shutting Down the Old Cylinder
With your buddy watching from the door—with emergency communications and their own SCBA within arm's reach—the first step is to close the valve on the empty cylinder. Turn it clockwise until it's firmly seated. Now, watch the system. The ejector is still running, creating a vacuum. You'll see the flow meter drop to zero as the system sucks all the remaining gas out of the line. This is a critical safety step to ensure there's no pressurized chlorine trapped in the line when you disconnect it.
3.3. The Most Common Mistake
Once the line is clear, you disconnect the yoke. Now, look inside the yoke. You'll see a small, soft lead washer. This is the single most important part of the entire process.
"Listen closely. More leaks are caused by this one tiny part than almost anything else. Never, ever reuse a washer. The old one has been crushed to make a seal; it won't seal properly a second time."
We discard the old washer and replace it with a brand new one. This is the number one cause of minor leaks, and it's completely preventable.
3.4. Connecting the New Cylinder
Bring the full cylinder into position. The very first thing you do—before you even think about taking off the valve protection hood—is secure the cylinder with chains. A 150-lb cylinder that falls over can shear its valve off. The escaping gas would turn that steel cylinder into an uncontrolled projectile capable of punching through a brick wall. You'll also notice a small plug in the valve. That's made of a special metal that will melt and release the gas if the cylinder gets too hot, preventing a catastrophic explosion. That's why we never store cylinders near steam pipes, heaters, or in direct sunlight. Only after it's chained do we remove the hood and inspect the valve face.
3.5. The Leak Test: A Moment of Truth
With the new washer in place, we connect the yoke and tighten it. Now, for the moment of truth. We perform a two-part leak test.
3.6. Going Live: The Quarter-Turn Rule
The connection is secure. Now we open the valve to put the cylinder in service. Watch how far I open it: just one-quarter to one full turn. That's it. This provides 100% of the gas flow the system needs.
Why not open it all the way? In an emergency, you need to shut this valve fast. A valve opened only this much can be closed in a single second. A fully opened valve takes precious time to close—time you don't have. Finally, the wrench always stays on the valve stem. It serves as a visual reminder that the cylinder is active and allows for immediate shut-off.
And that's a perfect change-out. But our job is also to be prepared for the day it isn't perfect.
If we ever have a real leak, your instincts might betray you. There are two rules you must burn into your memory because they are completely counter-intuitive.
As you can see, chlorine safety isn't a list of arbitrary rules. It's about deeply respecting the predictable laws of physics and chemistry.
Every protocol we follow is a layer of defense learned from experience. The buddy system respects toxicology. Using a new washer respects material science. Opening the valve just one turn respects ergonomics and reaction time. We do these things not just because a manual says so, but because the science dictates it is the only safe way. Chlorine is an unforgiving element, but it is a predictable one. Our job is to know its nature and act accordingly, every single time. Your life, and the lives of everyone here, depend on it.